Atom
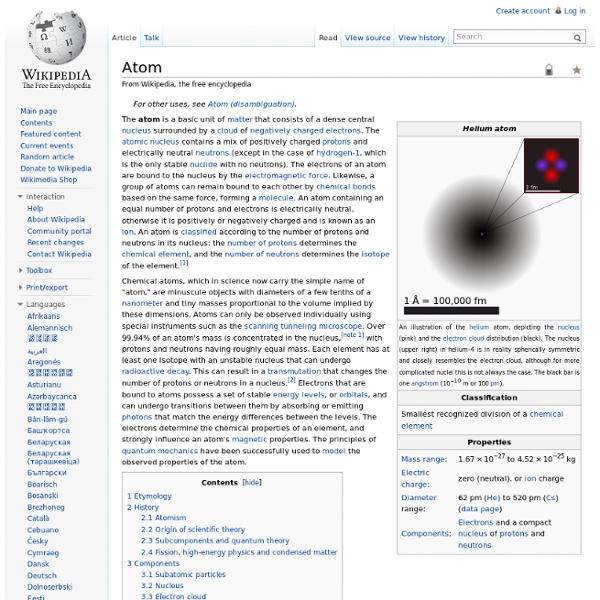
File:Atom diagram.png
Atom Diagram
Want to stay on top of all the space news? Follow @universetoday on Twitter A simple carbon atom. The image on the left is a basic atom diagram. This one shows the protons, neutrons, and electrons of a carbon atom. Each is in a group of six. Scientists have used atomic diagrams to explain the workings of the world for centuries. Basic chemistry explains the atom best. Earlier, I mentioned that there had been many atom models developed. The atom diagram is under constant revision as science uncovers more information about sub-atomic particles. Sources:WikipediaChemistry Help Tagged as: atom diagram
The Atom
As of July 1, 2013 ThinkQuest has been discontinued. We would like to thank everyone for being a part of the ThinkQuest global community: Students - For your limitless creativity and innovation, which inspires us all. Teachers - For your passion in guiding students on their quest. Partners - For your unwavering support and evangelism. Parents - For supporting the use of technology not only as an instrument of learning, but as a means of creating knowledge. We encourage everyone to continue to “Think, Create and Collaborate,” unleashing the power of technology to teach, share, and inspire. Best wishes, The Oracle Education Foundation
Fundamental Particle Structures
"Fundamental" Particle Structures Introduction It is customary in science, after discovering some kind of order or pattern, to form a hypothesis and test it by experiment. Because particle physics is not my area of expertise, I was unable to determine an appropriate experiment, but the pattern presented here is impressive in its implications for fundamental particle structures. By publishing the material, I hope to bring it to the attention of those more knowledgeable in this area, and that someone else may develop appropriate experiments to test my hypothesis. Some Background Electrons and protons, primary constituents of atoms, were both "discovered" (i.e., some of their properties were measured) in the late 1890’s. Another type of particle that began turning up (starting with the positron in 1932) was the anti-particle, so-called anti-matter. As particles were discovered, they were put into one of three groups: Hypothesis The picture of particle physics has been confused. neutrino. .
Related:
Related:



