

Mass testing for COVID-19 - The Lancet Microbe. When COVID-19 struck Europe, Slovakia was quick to respond.
It locked down early. Its population readily adopted face masks. At the end of April, 2020, Slovakia had the lowest COVID-19 death rate on the continent. But the second wave has proved harder to manage. On Sept 1, Slovakia had registered just over 4000 cases of COVID-19. On Oct 31–Nov 1, Slovakia tested 3·6 million of its 5·5 million inhabitants using lateral flow tests, the results of which are available within an hour.
“Our primary goal was to identify the people who were most infectious, the ones who had a high viral load and were the greatest epidemiological risk”, explains Pavol Jarčuška, associate professor in the department of infectology and travel medicine at PJ Šafárik University (Košice, Slovakia) and member of the Government's scientific advisory committee. The first weekend yielded 38 359 positive results, which translates to a disease prevalence of 1·06%. Article Info Publication History. 20-11-06, Good overview on rapid testing technology, Nature, New SARS-CoV-2 rapid tests, which can deliver results in a matter of minutes in the field rather than taking days in clinical diagnostic labs, are starting to become widely available.
They are based on a variety of technologies and formats, both old and new: conventional antigen-based tests for detecting viral proteins; polymerase chain reaction (PCR) assays for detecting viral genomic material; and newer CRISPR-based diagnostic tests, which also detect viral genomes but do so in a less complicated fashion. Rapid tests are easy to use: they can be deployed in high-risk environments, such as care homes or educational institutions, and by personnel with little or no specialist training. Some will be suitable for home use as well, once given the go-ahead by regulators. The need is urgent.
In recent weeks, numbers of confirmed cases have started to climb sharply in many countries. Christian Drosten: "Wir haben es selbst in der Hand" Rethinking Covid-19 Test Sensitivity — A Strategy for Containment. Global partnership to make available 120 million affordable, quality COVID-19 rapid tests for low- and middle-income countries. A set of agreements to make available, for low and middle-income countries, affordable, high-quality COVID-19 antigen rapid tests were today announced by the Access to COVID-19 Tools (ACT) Accelerator.
Organizations involved in the milestone agreement include the Africa Centres for Disease Control and Prevention (Africa CDC), the Bill & Melinda Gates Foundation, the Clinton Health Access Initiative (CHAI), the Foundation for Innovative New Diagnostics (FIND), the Global Fund, Unitaid, and the World Health Organization (WHO). As part of this comprehensive, end-to-end effort, the Bill & Melinda Gates Foundation has executed separate volume guarantee agreements with rapid diagnostic test (RDT) producers Abbott and SD Biosensor. These two arrangements will make available to LMICs 120 million antigen rapid diagnostic tests (Ag RDTs) – priced at a maximum of US$5 per unit – over a period of six months. Andrea F. About the ACT-Accelerator. Great article on antigen testing: Fast coronavirus tests: what they can and can’t do.
The United States leads the world in COVID-19 deaths but lags behind many countries — both large and small — in testing capacity.
That could soon change. At the end of August, the US Food and Drug Administration (FDA) granted emergency-use approval to a new credit-card-sized testing device for the coronavirus that costs US$5, gives results in 15 minutes and doesn’t require a laboratory or a machine for processing. The United States is spending $760 million on 150 million of these tests from health-care company Abbott Laboratories, headquartered in Abbott Park, Illinois, which plans to ramp up production to 50 million per month in October. The tests detect specific proteins — known as antigens — on the surface of the virus, and can identify people who are at the peak of infection, when virus levels in the body are likely to be high.
Proponents argue that this could be a game changer. Coronavirus: Die wichtigsten Testverfahren im Überblick - DER SPIEGEL. WHO COVID 19 lab testing 2020.1 eng. Why does the coronavirus test cost Rs 4,500 in India? On April 8, the Supreme Court of India directed the Central government to make testing for the novel coronavirus disease free of cost at approved private laboratories.
Whether the government should reimburse the labs for the cost incurred was a question that the court said it would consider later. Days later, on April 13, the court amended its order: free tests in private labs only for people covered by the government’s flagship healthcare programme Ayushman Bharat, and any other category of economically weaker sections of the society as notified by the government, it ruled. Private labs, the court clarified, could continue to charge for Covid-19 tests from individuals who could afford them. The price, however, could not exceed the upper limit set by the Indian Council for Medical Research: Rs 4,500. Almost everything related to testing in India, starting from whom to test to orders placed by the ICMR to procure test kits, have been subjects of heated debate. What explains the price gap? Corona-Tests: Testlabors in Containern sind bei Konzernen gefragt. Corona-Tests: Hilft viel testen wirklich?
Faktenpapier Testen. Test para coronavirus: ¿cómo es la prueba rápida y de US$8 que crearon en Argentina para detectar covid-19? Derechos de autor de la imagen María Eugenia Cerutti / Presidencia de la Nación La carrera por desarrollar pruebas, tratamientos y vacunas para la enfermedad covid-19 producto del coronavirus SARS-CoV-2 está en marcha desde hace meses.
Científicos de todo el mundo trabajan a contrarreloj para hallar soluciones para combatir esta enfermedad que ya se cobró la vida de más de 335.000 personas y registra más de 5 millones de infectados confirmados en el mundo, según el conteo de la Universidad Johns Hopkins al 22 de mayo. Una de esas soluciones se gestó en Argentina y es un kit de prueba "rápido y económico" que fue creado por un equipo de investigadores locales del sector público y privado.
Covid-19 test result delays are ruining America’s chances of stopping the coronavirus. In late June, an Austin, Texas, man with a runny nose and sore throat got a Covid-19 test and was told it could take up to 10 days for his results to come back.
While he waited, he shrugged off his symptoms as a cold and continued with his plans, which included attending a wedding outside Dallas. Several days after the wedding, however, he felt much worse, with shortness of breath and a cough. Serology testing in the COVID-19 pandemic response - The Lancet Infectious Diseases. 20-08-04, US, ‘It’s Like Having No Testing’: Test resuts delayed, NYT. “Right now, we live hand to mouth,” he said of the chemicals.

“I need a shipment on Monday to make sure all of my labs run through Wednesday, and then on Wednesday, I need another shipment,” he said, adding, “That’s how tight the supply chain is.” The six-state agreement announced on Tuesday showed how the lack of a federal testing program has left municipalities and states to fend for themselves. The Trump administration has provided new support to hard-hit regions by providing free coronavirus testing in cities through a “surge testing” program announced last month. But the bulk of government-sponsored testing has been provided by cities, counties and states that hire third-party contractors such as Quest and LabCorp. As a result, the length of the delay varies between states, and within them.
WHO head: 'Our key message is: test, test, test' News BBC News Navigation Sections Media player Media playback is unsupported on your device.
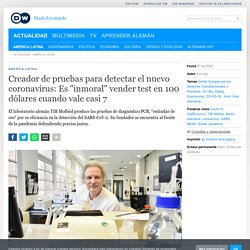
Creador de pruebas para detectar el nuevo coronavirus: Es ″inmoral″ vender test en 100 dólares cuando vale casi 7. "Visto en retrospectiva, esta pandemia era algo que ya sabíamos que podía suceder.
The explosion of new coronavirus tests that could help to end the pandemic. The timing couldn’t have been worse. In March, just as Thailand’s coronavirus outbreak began to ramp up, three hospitals in Bangkok announced that they had suspended testing for the virus because they had run out of reagents. Thai researchers rushed to help the country’s clinical laboratories meet the demand. Coronavirus Testing the Cheap, Simple Way. Simple at-home tests for the coronavirus, some that involve spitting into a small tube of solution, could be the key to expanding testing and impeding the spread of the pandemic. The Food and Drug Administration should encourage their development and then fast track approval.
One variety, paper-strip tests, are inexpensive and easy enough to make that Americans could test themselves every day. You would simply spit into a tube of saline solution and insert a small piece of paper embedded with a strip of protein. If you are infected with enough of the virus, the strip will change color within 15 minutes. Your next step would be to self-quarantine, notify your doctor and confirm the result with a standard swab test — the polymerase chain reaction nasal swab. - Coronavirus SARS-CoV-2 - Nationale Teststrategie – wer wird in Deutschland getestet? FDA, Coronavirus Testing Basics, great summary. Español You’ve probably heard a lot about coronavirus testing recently. If you think you have coronavirus disease 2019 (COVID-19) and need a test, contact your health care provider immediately.
The FDA has been working around the clock to increase the availability of critical medical products, including tests for the coronavirus, to fight the COVID-19 pandemic. Learn more about the different types of tests and the steps involved. Types of Tests There are two different types of tests – diagnostic tests and antibody tests. A diagnostic test can show if you have an active coronavirus infection and should take steps to quarantine or isolate yourself from others. Investigation of SARS-CoV-2 outbreaks in six care homes in London, April 2020 - EClinicalMedicine. Methods We assessed SARS-CoV-2 positivity in the residents and staff (carers and those without caring duties), recorded their symptoms in the two weeks prior to sampling and followed them daily for new symptoms and outcomes for two weeks through daily phone-call and datasheet completion. Care home data were collected systematically covering resident demographics, facilities, staffing and infection control measures in place at the time of swabbing (Supplement Table 2).
Staff working at the care home during the days of testing took nasal swabs for the residents and submitted their own samples by self-swabbing with appropriate instructions. Symptom status at the time of testing (symptomatic or asymptomatic) was recorded on the sample submission form. Laboratory methods Swabs from all six homes were couriered to the PHE reference laboratory on the day they were collected. Read later. Was Sie über Corona-Schnelltests wissen müssen. Coronavirus: Was kann der Antigen-Schnelltest von Roche? - DER SPIEGEL. Nach Ankündigung von Roche: Experte: Neue Corona-Schnelltests nicht absolut zuverlässig. Fast coronavirus tests: what they can and can’t do.
(29) Coronavirus-Update: Tests gezielter einsetzen. Sendedatum: 07.04.2020 13:15 Uhr Für viele beginnt der Tag ja mit der Zahl der gemeldeten Neuinfektionen. 20 04 07, (29) Coronavirus Update Tests gezielte...tzen NDR.de Nachrichten NDR Info. (31) Coronavirus-Update: Eine Wiederinfektion bleibt unwahrscheinlich. Sendedatum: 14.04.2020 14:00 Uhr. (39) Welche Chancen neue Tests bieten. (39) Coronavirus Update Welche Chancen...eten NDR.de Nachrichten NDR Info. Christian-Drosten-Podcast: Was gibt es Neues zum Coronavirus - DER SPIEGEL. Zweite Corona-Welle: Ein Plan für den Herbst.
20-07-10, Coronavirus: Wird in den USA einfach nur mehr getestet? - DER SPIEGEL. 20-08-03, Coronavirus: Was wir über die neuen Schnelltests wissen - und was nicht - DER SPIEGEL. In-depth reporting on science and technology. There are no drugs or vaccines for the new coronavirus. It will take at least 18 to 24 months before an appropriate vaccine can be developed, tested for side effects in all test phases, approved by the health authorities, and then produced in large quantities and delivered globally. And then people will have to be vaccinated.
As such, reliable tests in the coming months are the most sensible measure to at least slow the further spread of the virus. Tests can be used to identify infected people and possible "hot spots. " This is the only way to quarantine those infected — whether in a medical facility or, in the case of a mild course, isolated at home. However, even in well-developed countries, medical facilities and laboratories quickly reach the limits of their capacity during an acute wave of infection. Who is tested? According to current estimates, simply being in a risk area is not enough to justify a test. Economy and finance news from a German perspective. If you are only coughing, that's not enough. If you want to be tested for SARS-CoV-2, as a rule, you not only have to have symptoms, but you also have to have been in contact with someone who was infected. These are the guidelines of the Robert Koch Institute, Germany's central authority for the identification, surveillance and prevention of infectious diseases.
Coronavirus: What you should know about tests. More and more tests for the novel coronavirus are coming onto the market. By the end of April, there were already more than 150 different products worldwide. These can essentially be divided into two main groups: tests that prove that someone is carrying the coronavirus in them and can therefore potentially infect others, and serological tests that prove that someone has been infected in the past. Detection of acute infection To check whether someone is currently infected and can also infect others, a test based on the polymerase chain reaction (PCR) method is usually used.
Delay in testing is critical - The Lancet Public Health. Summary. 20 04 18, Sensitivity of Rapid Tests, Elsevier. 20 04 02, SARS by the numbers, elife. 20 03 19, COVID 19 laboratory 2020.5, Guidelines, WHO eng. 20 03 21, WHO COVID 19 lab testing 2020.1, WHO eng. 20-07-14, Unterhaltung mit Felix über Tests: